
The heart is hollow elastic muscular pump which pumps blood to the whole body to meet the metabolic demands. Briefly reviewing the circulation, first the de-oxygenated blood from the veins enters into the right atrium which is pumped by RV to the lungs for oxygenation. The oxygenated blood enters left atrium and upon opening of the mitral valve it enters the LV when it is relaxing (LV diastole). The blood from the left atrium begins to fill it due to pressure gradient. When LA & LV have an equal amount of blood and its pressure (Diastasis) then LA contraction (LA kick) occurs and that forces more blood (20-25%) in to the LV. Following this the LV systolic phase begins with ejection of stroke volume to the systemic circulation.
For many years LV systole was considered as active energy dependent process while the diastole was considered as a passive process. Lusitropy refers to the ability of the myocardium to relax following excitation contraction coupling. Lusitropy is diastolic function while inotropy (or contractility) is systolic function.
Historical Aspects : Galen in 100 B.C. first proposed that the heart is filled by RV dilatation. In 1628, William Harvey recognized that the heart was the central pump in a circulatory system containing arteries and veins. This helped to understand that the CHF was caused by damage or weakening of the heart muscle and a decrease in LV pumping function.
The Diastole was ignored and was just thought as the passive relaxation of LV following the systole. Frank-Starling mechanism demonstrated how LV stroke volume was regulated by LV end diastolic volume and LV contractility. Ultimately, it became clear that like LV systolic dysfunction, the LV diastolic dysfunction alone could cause symptoms and those diastolic and systolic LV functions were interrelated. Katz observed that the ventricles actually act as suction pump after mitral valve opening in mammalian heart. It was also recognized that a limited filling capacity was the cardinal feature of constrictive pericarditis that reduced cardiac output and resulted in marked peripheral edema and generalized wasting.(1)
Physiology and pathophysiology of LV relaxation : Zile and Brutsaert (2) defined LV relaxation as ‘the time period during which the myocardium loses its ability to generate force and shorten and returns to an unstressed length and force’. From a mechanical point of view, the transition between systole and diastole was described as the time of aortic valve closure. However, the transition between contraction and relaxation corresponds to the dissociation of actin–myosin cross-bridges that begins during the early phase of LV ejection, before aortic valve closure. The figure shows all the metabolic process going in the cardiac myosite.

Fig. 1 –
Following the lowering of intra-cellular Ca++ concentration the dissociation of actin–myosin cross-bridges occurs. This transfer of calcium from the cytosole into the sarcoplasmic reticulum (SR) requires energy. Phosphorylation of phospholamban that is needed to activate the ATPase-induced calcium sequestration into the SR, Na+/ Ca++ exchanger-induced extrusion of Ca++ from the cytoplasm, release of Ca++ from troponin C, detachment of the actin–myosin cross‐bridges, and return of the sarcomere to its resting length are all energy-consuming processes. Thus, alterations leading to diastolic dysfunction may involve phenomena that occur not only during ‘classic’ diastole, but also earlier in the cardiac cycle, at the time when intracellular calcium falls.
Calcium-induced calcium release in cardiac myocytes : After electrical stimulation, calcium influx through the calcium channels (Ica) stimulates ryanodin receptors (RyR) to release more calcium from the sarcoplasmic reticulum (SR) into the cytosol. Calcium concentration falls (i) by reuptake in the SR via phospholamban (PLB) stimulation and (ii) by calcium efflux by Na+/ Ca++ exchange (NCX).
Functional factors are likely to occur during the peri-operative period or in the ICU. Affecting LV relaxation, they may precipitate latent LV diastolic dysfunction.
Contraction–relaxation coupling : LV relaxation begins early during systole indicating that relaxation and contraction are intimately coupled. The loss of atrial contraction associated with atrial fibrillation not only alters LV filling but also results in a slowing of myocardial relaxation.
Several other factors known to alter contractile function, including changes in afterload and the use of inotropes, markedly affect relaxation. However, the effect of preload variations on relaxation is not clear.
Components of Diastole :- Few facts : Pre-load of LV is LV end diastolic volume and Ventricular diastole is an active energy consuming process and hence blood flow passing through the coronary vasculature through LV muscle mass is determined by LV muscle relaxation and LVEDP along with normal coronary vasculature and aortic diastolic blood pressure.
1) Isovolumetric relaxation: This phase of cardiac cycle begins when aortic valve closes and mitral valve is yet to open. Thus the ventricles are relaxing without any without any change in ventricular blood volume but with rapid decline in intra-cavity pressure. The Isovolumetric relaxation time (IVRT) is prolonged in any condition that impairs active relaxation (e.g. myocardial ischemia). IVRT is shortened by rise in LA pressure which causes early opening of the mitral valve.
2) Early Rapid filling: This begins with the opening of the mitral valve and since the ventricles are still relaxing rapid filling due to pressure gradient and accounts for 75-80% of the ventricular filling. Early diastolic filling can be characterized (invasively) by the rate of volume change of the LV, the dV/dt
3) Diastasis: Once the pressure between LA and LV equalizes due to LV filling and LA emptying the flow across open mitral valve decreases resulting in this phase. The main determinant of the LV filling at this time is the LV chamber compliance (dV/dP) or LV chamber stiffness (dP/dV) and is dependent on the following factors like intrinsic myocardial stiffness, LV mass and volume, pericardial stretching, preload and RV volume (Ventricular interdependence)
4) Atrial systole: The active LA contraction (“atrial kick”) responsible for the 20-25% filling of LV and increases the trans-mitral gradient. This contribution is important and can increase even up to 35 % where LV relaxation (aortic stenosis) is hampered.
Left Ventricular Diastolic Dysfunction (LVDD) : LVDD refers to abnormalities of diastolic distensibility, filling, or relaxation, regardless of whether LV ejection fraction (LVEF) is normal or abnormal and whether the patient is symptomatic or not. The abnormality may be either reduced ventricular compliance (distensibility) or poor ventricular relaxation (reduced lusitropy). The term diastolic heart failure has been termed Heart Failure with normal Ejection Fraction (HFnlEF) to contrast it with LV systolic heart failure which has reduced EF (HFrEF).
Prevalence & Determinants of Diastolic Dysfunction in a General Population (3)
The reported prevalence of diastolic dysfunction in these studies varied from 27.3 to 34.7%, and was influenced by a number of factors. Advancing age, Female sex, higher BMI, heart rate, systolic blood pressure, serum insulin and creatinine. In a cross-sectional study of participants of the Flemish Study on Environment Genes and Health Outcomes (FLEMENGHO), approximately 50% of hypertensive subjects had impairment of LV diastolic function, (12% oonly in normotensive subjects). The prevalence of HFnlEF in the heart failure population is about 50-55%. If signs and symptoms of congestive heart failure-CHF (effort intolerance, dyspnoea, and pulmonary oedema) develop in a patient with normal or near normal EF it may be diastolic heart failure.
Normal diastolic function is dependent on compliance, distensibility and relaxation properties of the ventricles. Examining the pressure volume loops provide data regarding diastolic function. First of all a detailed examination of the diastolic phase of the cardiac cycle is in order.

Fig. 2 –
Compliance: Compliance or distensibility is defined as the ratio of volume change to the corresponding pressure change or as the slope of the volume–pressure (dV/dP) relationship. Elastance or stiffness is the inverse of compliance (dP/dV). Decreased compliance or increased stiffness is thus defined as an increase in the steepness of the pressure–volume plot. 2 causes of poor diastolic compliance : Increased chamber stiffness: This occurs in aortic stenosis or systemic hypertension due to LVH. Increased muscle stiffness: This occurs in restrictive cardiomyopathies due to amyloidosis and hemochromatosis.
Distensibility: Decreased ventricular distensibility is defined as an increased diastolic pressure at a given volume. This would be represented in a diastolic pressure volume diagram by a parallel upward shift of the entire pressure–volume relation.
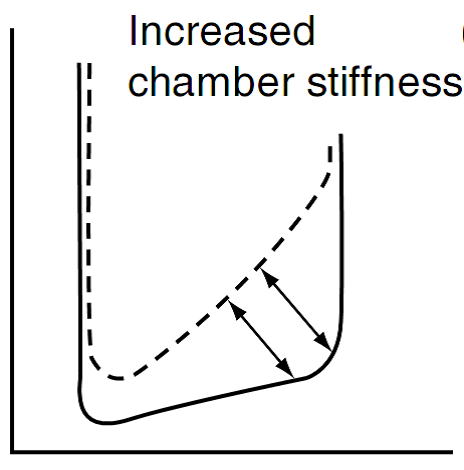
Fig. 3 –
Decreased distensibility can occur from intrinsic and extrinsic causes.
| Extrinsic Causes |
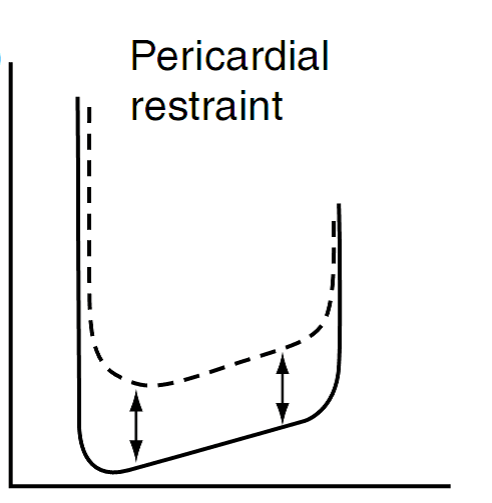
Fig. 4 –
( P/V loop in diastolic dysfunction due to reduced distensibility. The solid loop is the normal curve).
Intrinsic causes: Coronary artery disease is one of the important reasons for diastolic dysfunction. There is diminished distensibility which is seen even earlier than systolic dysfunction. For a given diastolic volume the diastolic pressure will be higher
Extrinsic causes: Decreased distensibility may be caused by extrinsic limitations to ventricular expansion in diastole. Diminished distensibility occurs due to ventricular interdependence and the restraining effect of the pericardium. For example, distension of the right ventricle with a leftward septal shift will result in diminished distensibility of the left ventricle. In addition, reduced distensibility may occur due to restrictive pericarditis or pericardial tamponade or fluid filled pericardium.
Relaxation is impaired during myocardial ischemia and in patients with hypertrophic and congestive cardiomyopathies.
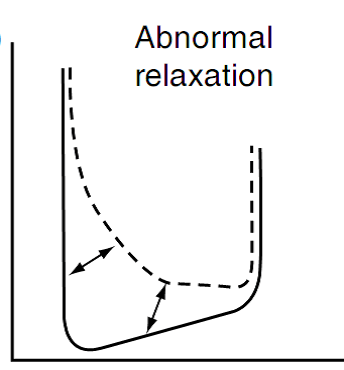
Fig. 5 –
Diastolic pressure volume relationship when the ventricular relaxation is impaired having LV abnormalities of LV diastolic function.
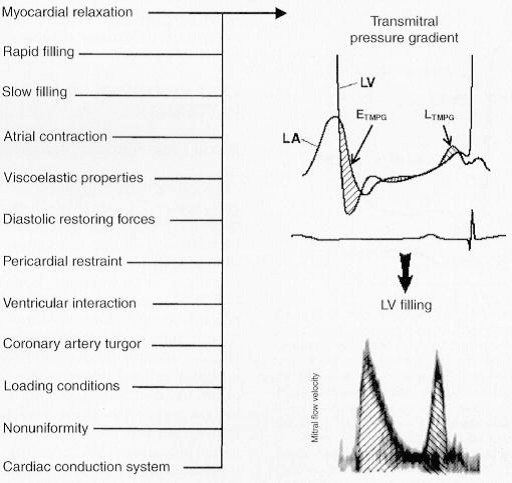
Fig. 6 –
(Numerous factors affect LV diastolic properties and filling; however, all factors work through their effects on the transmitral pressure gradient (TMPG), which is the actual determinant of LV filling. An early positive TMPG (ETMPG ) relates to early diastolic LV filling as assessed by pulse-wave Doppler mitral flow velocity. A reverse pressure gradient in mid-diastole decelerates early filling, with late filling (LTMPG ) occurring as a result of the increase in the left atrial pressure owing to atrial contraction)
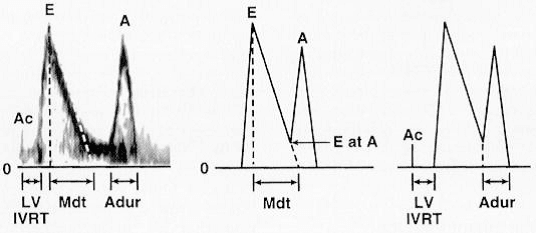
Fig. 7 –
(Pulsed-wave Doppler mitral flow velocity pattern illustrating the mitral flow velocity variables measured during a diastolic function examination. These include peak E-wave velocity, peak A-wave velocity, and E- at A-wave velocity. Time intervals measured include the LV isovolumic relaxation time (IVRT) (the interval from aortic valve closure to start of mitral flow). The mitral deceleration time (DT) is defined as the interval from peak E-wave velocity to the point on the velocity baseline obtained by linear extrapolation along the velocity deceleration slope. The A-wave duration (A dur) is measured from the E- at A-wave velocity to the end of mitral flow after atrial contraction. When E- at A-wave velocity is increased, a vertical line is drawn to this point as shown in the far right panel”) (1).
Clinical symptoms of Diastolic dysfunction : 1)Dyspnea on exertion or later at rest also is the main symptom.
2) Fatigue (Francis et. al 2000). “Chronic fatigue syndrome” is presenting symptom of LV diastolic dysfunction and failure. Classical CHF like symptoms like wheezing, coughing, edema feet. Gallop’, murmurs, ‘heave’ and lung sounds (‘rales’) (Francis et. al. 2000).
Features of Diastolic heart failure with Altered versus Preserved E.F.
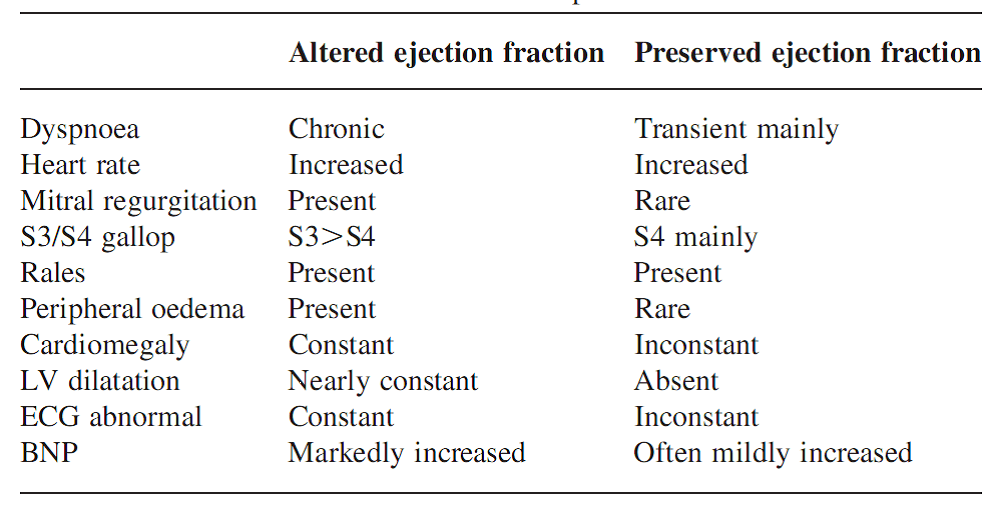
Fig. 8 –
Diagnosing diastolic dysfunction: Laboratory tests : BNP levels are raised in presence of heart failure
EF is not a reliable method to assess LVDD. Left ventricular end diastolic pressure (LVEDP) – can reliably determine diastolic dysfunction (Zile et. al. 2001) by Cardiac catheterization. Disadvantage is it is an invasive and expensive procedure.
ECHOcardiography is a simpler investigation.- 2-D echo, pulsed-wave Doppler, M-mode colour Doppler, and tissue Doppler imaging
- Mitral inflow patterns : E/A, decceleration time, IVRT
- Mitral annulus velocities on tissue doppler: E/e’ ratio
- Pulmonary venous inflow patterns
E/A ratio : The flow from the left atrium to the left ventricle occurs in 3 phases: The ‘transmitral velocity’ (measured by E and A waves) and deceleration are 2 important measures. The LV filling occurs in two phases; first the passive blood flow from full LA to almost empty LV during relaxation phase produces ‘E’ wave (Early rapid filling), a period of low or no flow (Diastasis), while ‘A’ wave is the wave of LA contraction forcing blood in LV. While these waves can be analyzed by studying the movement of the anterior mitral leaflet in M-mode, it is best done with Pulsed wave Doppler (PWD). The PWD cursor is placed between the tips of the open mitral leaflets in the Apical 2 Chamber or Apical 4 Chamber views.
Decceleration time (Dct) : Mitral inflow wave pattern (PWD), downslope from peak of E wave is measured to calculate Dct. Dct is dependent on a pressure gradient from LA to LV. Dct increase as LV relaxation decreases.
E/e’ ratio : Tissue doppler (TDI) is used to calculate diastolic peak velocities of the mitral annulus. These peak velocities are named e’(medial)- 2-5 mm sample volume over medial mitral annulus and e’(lateral) 2-5 mm sample volume over lateral mitral annulus in Apical 4 chamber view
Pulmonary vein inflow – Pulsed doppler cursor at the entry of pulmonary veins in to LA in Apical 4 C view is placed and PWD trace is obtained and frozen. Normally biphasic pulmonary vein flow with dominant systolic, S wave and less dominant diastolic D wave is seen.
Isovolumic Relaxation time (IVRT, IRT) : IRT measures the time from the closing of the aortic valve till ventricles relax and mitral valve opens. Healthy hearts usually take about 70 +/- 12 ms (microseconds) while hearts with diastolic dysfunction usually take about 110 ms to fully relax.
Atrial Flow Reversal : Even in healthy people, the force of the atrial contraction causes some blood to go back into the pulmonary veins and is called an Ar wave. As left ventricular pressure builds up in heart failure due to increased stiffness of the ventricles the atria have to contract forcefully. In diastolic dysfunction, the amlitude of the A-wave is over 25 cms. and its duration lasts longer than that of the A wave.
LVDD : Stages
(1) Reduced relaxation stage – In this first stage of diastolic dysfunction the left ventricule is unable to relax enough to accept normal amounts of blood during the first (E) phase of ventricular filling. A reduced relaxation phase manifests itself as reduced E/A wave ratio.
An E/A wave ratio of less than 0.75 is considered to be indicative of diastolic dysfunction. Some controversy exists, however, how important a low E/A ratio, are. Because only a small fraction of people (2-3%) with a low E/A ratio have signs or symptoms of heart failure a low E/A ratio is not necessarily evidence of significant diastolic dysfunction or diastolic heart failure.
(2) Pseudonormal stage– As the diastolic dysfunction increases proceeds the E/A ratio normalizes; i.e. achieves a pseudonormal appearance (between .75 and 1.5). This occurs when the increased stiffness of the left ventricule inhibits blood flow during the later phase (A wave). However, pseudonormal can be differentiated from normal by 1) presence of enlarged LA on , 2) Ar wave >25 cm/sec with prolonged duration and 3) and advanced NYHA (IIa to NYHA III) category symptoms.
(3) Restrictive stage – In this stage, as LV enlargement and fibrosis prevents the left ventricle from expanding during the atrial contraction phase, the E/A ratio reverses and a high E/A ratio is seen (>1.5) and no amount of atrial contraction can fill it and hence LV receives blood during the first (relaxation) phase.
| GRADES Of Diastolic Dysfunction | E/A RATIO |
| Normal | Between .75 & 1.5 |
| 1 (Reduced relaxation) | < 0.75 |
| 2 (Pseuedonormal) | Between 0 .75 and 1.5 |
| 3 (Restrictive) | > 1.5 |
Based on the information given above a generalized chart can be developed (Koren 2002, Galderisi 2005, Pollard and Pozzoli 2001).
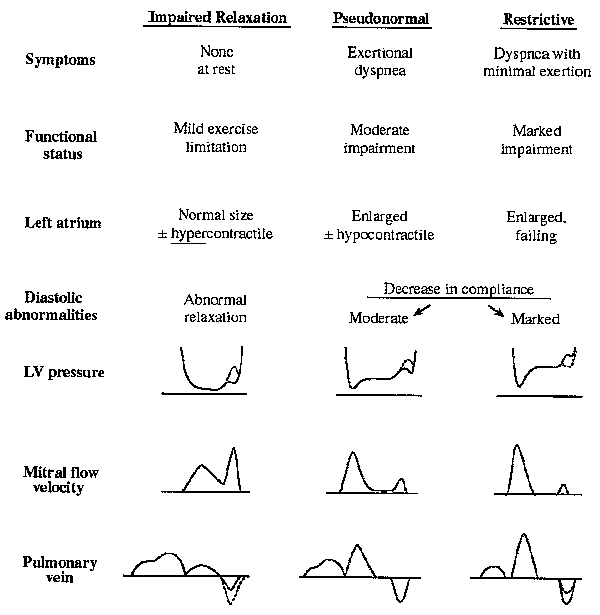
Fig. 9 –
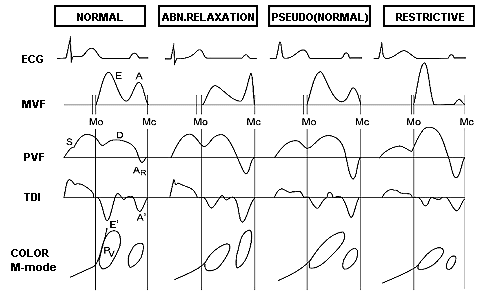
Fig. 10 –
| Degree of dystolic dysfunction | E/A | IVRT | E wave deceleration | Atrial reversal | Ar vs. A wave |
| >1 | *70 +/- 12 | 200 | <25 | Smaller | |
| Mild | <1 | >100 | >240 | <.35 | Smaller |
| Mild to moderate | <1 | >100 | >240 | >.35 | 20 ms > |
| Moderate | (Pseudo)normal 1-2 | >100 | ? | >.35 | 25 ms >s |
| Severe (restrictive) | >1.5 | >60 | <150ms | >.25 | ? |
*> 40 years of age = 80 +/- 12

Fig. 11 –
Markers of elevated LV filling pressure:
E/A > 2
Dct < 160ms
E/e’ (medial) >15
E/e’ (lateral) >10
PFV S/D <40%
PFV AR amplitude >25 cm/sec
PFV AR duration > 30ms more than A wave
LA enlargement
LV hypertrophy
Formulae to calculate LA pressure
Sinus rhythm
2+1.2(E/e’)
Sinus tachycardia
1.5+1.5(E/e’)
Atrial fibrillation
6.5+0.8(E/e’)
The E/e’ included in the above calculations indicates that obtained from the medial mitral annulus.
Treatment of Severe Diastolic Dysfunction : ACE Inhibitors – enalapril, lisinopril) and angiotensin receptor blockers(ARB’s – candesartan, losartan) are among the most common pharmaceuticals used in treating diastolic dysfunction. They reduce Hypertension, LVH, LV remodelling and improve quality of life.
Beta blockers (atenolol, metoprolol) are the other main treatment for diastolic dysfunction. By slowing the heart rate BB’s cause increased left ventricular filling time. They also have anti-hypertensive action and inhibit rennin release.
Calcium channel blockers – Reduce LVH and improve symptoms.
Diuretics – In acute setting of heart
Anaesthesia and the significance of mild diastolic dysfunction :
Should we worry about presence of LVDD in patients posted for surgery ?
313 patients undergoing vascular surgery were studied. 8% (n=24) of patients had isolated systolic dysfunction, 43% (n =134) had isolated diastolic dysfunction, and 24% (n=75) both systolic and diastolic dysfunction. The most common postoperative adverse outcome was CHF 20% (n= 62) and prolonged ICU stay. Age, Vp (tran-mitral propagation velocity), type of surgery, female gender, and renal failure were predictive of postoperative adverse outcome (7).
77 patients undergoing CABG surgery were divided in 3 groups: 32 (n= Normal LV relaxation), 38 (AR=Abnormal Relaxation) and 7 (PN=Pseudo-normalization group). In groups where LVDD was present patients became haemodynamically unstable after induction as comared to normal group. (8).
Other stdies also have proved that mild or chronic LVDD causes cardiac dysfunction and should be assessed pre-operatively. (9) (10).
Pre-operative assessment of patients of LVDD at risk of developing Diastolic heart failure : Careful history taking about Chronic fatigue syndrome, NYHA grading of breathlessness, Inability to perform activity < 4 METs (climbing a flight of stairs) is associated with high risk. Elderly persons, H/o edema, presence of co-morbidities like long standing systemic hypertension, Aortic stenosis, Diabetes, Coronary artery disease, H/o of diastolic heart failure, arrhythmia, increased BMI, Obesity, CHF are few indicators to further evaluate these patients for the possible diastolic dysfunction.
Echo : Routine screening for Cardiac chamber dilatation, hypertrophy, Valvular diseases, pericardial fluid. E/A ratio’s less than 0.6 (= impaired relaxation, mild diastolic dysfunction) were not associated with increased mortality in an older population.
High E/A ratio’s (>1.5), however, were associated with an increased risk of mortality (Bella et. al. 2002). Another five year follow up study found mild disastolic dysfunction conferred a significantly increased risk of mortality (Redfield et. al. 2003). This study, however, accounted for many fewer confounding factors that the Strong Heart Study. These studies suggest findings of restrictive filling may place someone with few symptoms of heart failure at risk.
Peripheral and minor procedures : Local or Regional anaesthesiaIntraoperative Management
G.A. – Goals are preserving Cardiac output and reducing myocardial work. During the perioperative period the haemodynamic changes affecting diastolic time, such as tachycardia, arrhythmia and myocardial ischaemia, are likely to decompensate further pre-existing diastolic dysfunction.
Proper selection of Induction agents like etomidate, Fentanyl, muscle relaxants, Inhalational agents like Isoflurane, Sevoflurane, Narcotics are safe agents.
Preseving Cardiac Output : 3 factors influencing Cardiac Output are Preload, Contractility and Afterload. LV be adequately filled during diastole. More than normal CVP, avoidance of tachycardia and arrhythmia. Maintenance of Sinus rhthm for the preservation of “Atrial kick”. Contractility is maintained by Dobutamine. Afterload is judiciously reduced but avoid overzealous reduction to prevent myocardial ischemia.
Avoid Myocardial ischemia : Avoid hypertensive episodes and hypotension, treat tachycardia
Sympathetic tone should be maintained as anaesthesia induction may reduce the sympathetic tone and hypotension may occur. Judicious doses of ephedrine are given.
Adequate pain relief, avoidance of hypoxia, hypercabia, intubation and extubation stressors are avoided. GA with epidural anaesthesia with post operative epidural analgesia helps in reducing stress and tachycardia.
Intra-operative monitoring : Invasive arterial pressure, CVP, PCWP, TEE, Cardiac output monitoring should be point of care in all major cases.
Post-operative Care : Supplemental oxygen therapy, Fluid balance, Good post operative pain relief, adequate monitoring and ICU care are all important. Treat anaemia, hypoxia, Shivering, hypothermia, Acid-base and electrolyte derangements and blood sugar control. Inotropes and vasodilators like NTG help in maintenance of haemodynamics.
ACEI or ARB drugs in appropriate doses should be introduced early.
Features of heart failure including Diastolic Heart failure :
The Framingham criteria for diagnosis of heart failure. This requires 2 major and 1 minor criterion
Major criteria: Paroxysmal Nocturnal Dyspnea or orthopnea, Jugular venous distension of CVP > 16 mm Hg,
Rales or acute pulmonary edema, Cardiomegaly, Hepatojugular reflex, Response to diuretic (weight loss> 4.5 Kg in 5 days)
Minor criteria: Ankle edema, Nocturnal cough, Exertional dyspnea, Pleural effusion, Vital capacity < 2/3 of normal, Hepatomegaly, Tachycardia > 120 bpm..
Treatment for acute diastolic heart failure : 1) Diuretics, Nitrates 2) Hypertensive crisis control
3) Inotropes (Dobutamine, Milrinone, Levosimendan)
Conclusion : LV diastolic dysfunction (LVDD) is quite an underestimated and un-anticipated cardiac dysfunction. Many patients walking for routine surgery may be suffering from LVDD and careful history and high degree of suspicion in patients like Obesity (high BMI), Long standing hypertension, Diabetes, ischaemic heart disease, Valvular heart diseases, females. Dyspnea on exertion and Chronic fatigue syndrome. The patients are properly investigated accordingly. Normal EF does not rule out LVDD. Every attempt must be made to otimize the factors for smooth sailing in the peri-operative period and acute diastolic heart failure should be suspected early and treated accordingly.
Declaration : The information, diagrams, tables compiled in this article is obtained from several internet articles and I thank them for the invaluable information put on the search engine…..Dr S K Deshpande
References
- Christopher P. Appleton, Firstenberg et al : The ECHO-Doppler evaluation of LV Diastolic function, A Current Perspective, Cardiology Clinics, 2000, 18, Number 3
- Pirracchio1, B. Cholley – Diastolic heart failure in anaesthesia and critical care, Br. J. Anaesth. (2007) 98 (6):707-721
- Tatiana Kuznetsova; Lieven Herbots; Yu Jin; Katarzyna Stolarz-Skrzypek; Jan A Staessen Systolic and Diastolic Left Ventricular Dysfunction: From Risk Factors to Overt Heart Failure Expert Rev Cardiovasc Ther. 2010;8(2):251-258
- P. R. Suneel, Internet article on LVDD, Dept. of Anesthesia, SCTIMST, Trivandrum, India
- Tutorial 6, Assessment of LVDD– ICU Sonography,CMCH, Vellore, TN, India.
- EAE / ASE recommendations : Recommendation for the evaluation of LV diastolic dysfunction by Echocardiography – European Journal of Echocardiography (2009) 10, 165–193
- Robina Matyal, Philip Hess, Balachundhar Subramaniam et al, Perioperative diastolic dysfunction during vascular surgery and its association with postoperative Outcome, J Vasc Surg 2009;50:70-6
- Harumasa Nakazawa, Takeshi Nomura. Importance of Preoperative LV Diastolic Function Evaluation in Patients Undergoing Cardiac Surgery, Proceedings of the 2010 Annual Meeting of the American Society Anesthesiologists
- Amit Bardia, Jayant Jainandunsing, Left atrial Volume Index, an indicator of chronic diastolic dysfunction, SCA bulletin, 2012, 11:3.
- Conture P, Denault AY et al, Effects of anesthetic induction in patients with diastolic dysfunction. Can J Anaesth J 2009 May;56(5):357



