
Dr. Vrishali Ankalwar,
Associate Professor, Dept. of Anesthesiology,
GMCH, Nagpur
Background: Of all treatments in contemporary psychiatry, perhaps none is more commonly misunderstood than electroconvulsive therapy (ECT). Its depiction in movies such as One Flew Over the Cuckoo’s Nest has contributed to its controversial reputation in the general public. Electroconvulsive therapy (ECT) has mention in long back 80 years i.e. in 1930 in Italy but first successful use is documented in 1936 by Italian psychiatrist by Cerlitti and Bini in 1937 but very soon it became frightening experience for psychiatric patients.
Earlier medicine named Metrazol was used to induce seizures but soon replaced by electric shock. Beginning in the 1940s, the electrical technique was adopted by almost every major psychiatric institution around the world as a treatment for serious mental disease. Still, without anesthesia, upto 1950, ECT was used to threaten an unruly patient or to punish a violent patient at asylum. In 1960 – 70, antipsychiatry movement attacked psychiatry and ECTs. ECTs are considered inhuman and tortuous resulted in fading of use of ECTs. With advances in anesthesia including better induction agent lead to resurgence of ECTs in 1980s.
ECTs given without general anesthesia are termed as unmodified ECTs which used to be associated with musculoskeletal injuries in 40% patients. Most worrisome was post ECT vertebral fractures. ECTs done under general anesthesia are said as modified ECTs.
Today, it is highly sophisticated procedure and a widely accepted treatment for serious mental disorder and is taught and practiced at hospitals throughout the world. It is estimated that one million psychiatry patients receive ECT annually. In our own institute which is a tertiary care centre has annual 1000 ECTS per year. Still, damage to brain and awareness are the biggest fear amongst relatives of psychiatric patients.
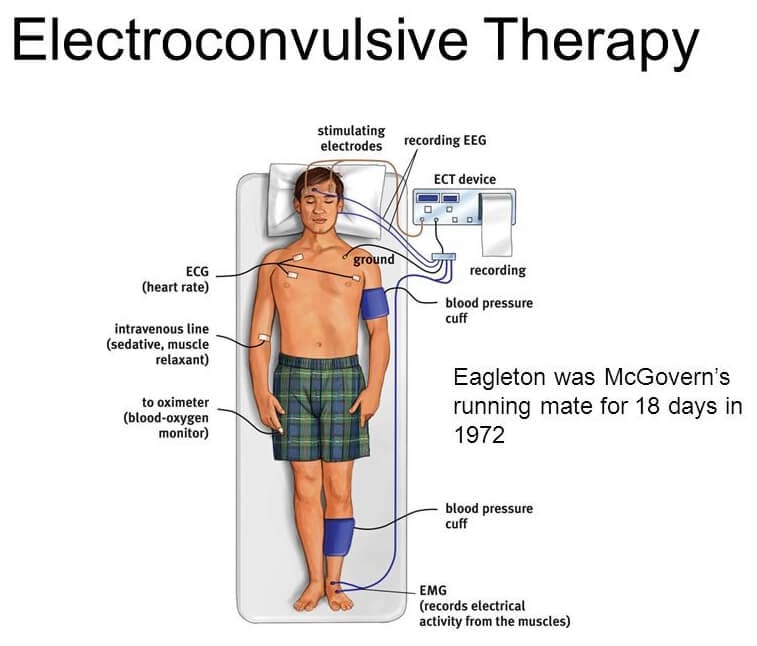
Physiological effects of ECT: ECT is the induction of a generalized seizure by electrical stimulation of one or both cerebral hemispheres. Simultaneously, it induces transient but significant cerebrovascular and cardiovascular changes. After brief pulse of ECT (1sec), there is great increase in cerebral blood flow and intracranial pressure. Cardiovascular effects result from autonomic nervous system (ANS) activation during ECT procedure. There is initial parasympathetic discharge of 10-15 sec manifested as bradycardia, occasional asystole, premature atrial and ventricular contractions, hypotension and salivation (tonic phase) which is followed by sympathetic discharge (clonic phase) also termed as ‘Catecholamine storm’ leads to tachycardia, hypertension and ECG changes, like ST segment depression and T- wave inversion can be seen and rarely ventricular tachycardia. This phase generally resolves within 5-10 min.
Considering these physiological changes and related complications, goals of anesthesia for a patient posted for ECT aim at providing amnesia and muscle relaxation but the level of anesthesia should not be so deep as to suppress seizure activity.
Anesthetic considerations for ECT:
Pre-medication with anticholinergic agents such as glycopyrrolate and atropine are often given to antagonize the initial parasympathetic discharge. Glycopyrrolate (0.01 mg/kg) is preferred and administered either intramuscularly at least 3 min prior the scheduled procedure or intravenously just before injecting the induction agent. It may decrease the likelihood and severity of bradycardia or asystole and the risk of aspiration due to vagal effects of ECT. Although routine use of anticholinergics has been criticized as unnecessary, we prefer its use in our institute.
Oxygenation – Cerebral oxygen consumption increases almost 200% during the seizure. It is, therefore, a standard recommendation that the lungs should be ventilated with 100% oxygen at a rate of 15–20 breaths/min, beginning approximately 1 min before the induction, continued until the resumption of spontaneous breathing.
Induction agent – Sodium pentothal (2–4 mg/kg) was the first induction agent used because of its availability at that time. Methohexital (0.5–1.0 mg/kg) is considered as the ‘gold standard’. However, unavailability at many centers limits its use. Ketamine (1.5–2 mg/kg) and Etomidate (0.15–0.6 mg/kg) might seem preferable to other agents in the light of their lack of anticonvulsant properties. However, other aspects such as drug’s safety require consideration.
Propofol, due to its shorter half-life with the advantage of minor haemodynamic effects, it is universally becoming the induction agent of choice, in spite of higher cost and anticonvulsant effect at higher doses.
Ketamine-propofol mixture (“ketofol”) and ketofol-dexmedetomidine combination (ketofol-dex mixture) can be used as alternative induction agents, which overcome disadvantages of individual agents. Ketofol used in 1:1 found to be very effective induction agent and preferred method in many centers including our center.
Sevoflurane (5%–8% for induction, followed by 1–2 minimal alveolar concentration [MAC]) is the only inhalational agent which can be opted for induction in ECT especially in non cooperative patients for IV access . Sevoflurane also can be used as an induction agent safely in the third trimester of pregnancy due to its tocolytic effect and for patients having history of coronary vascular disease. In patients with refractory to seizure, remifentanil can be used as sole induction agent.
There is no strong evidence to recommend a particular induction agent for ECT. They should be chosen on the basis of their effect on seizure quality, adverse effect profile and emergence time. Whichever drug is used, it is preferable to utilize the same one throughout a course of ECT and rarely may need to be changed during the course of treatment.
The goal of ECT is to induce a motor and a central EEG seizure of at least 25 and 40s respectively. Induction agents in the descending order of seizure duration after their use are:
Etomidate > ketamine > methohexital > sevoflurane > thiopental > propofol.
Muscle relaxation – Muscle relaxation is used to eliminate musculoskeletal injury and aid in airway management. Succinylcholine (0.5–1.5 mg/kg) remains the relaxant of choice due to its rapid onset and short duration. Atracurium, Mivacurium or Rocuronium may be acceptable alternatives, although their relatively prolonged action will need continued anaesthesia and/or active reversal after treatment.
To attenuate the sympathetic response of surge in systolic pressure and heart rate after ECT, beta blockers such as esmolol (1 mg/kg) and labetalol (0.3 mg/kg) can be used. However, they should be considered after a detailed evaluation of patient’s cardiovascular risk. Esmolol has a lesser effect on seizure duration than labetalol. Calcium channel blockers and alpha 2 agonists can also be used but rarely needed.
Role Of Induction Agents In Potentiating Therapeutic Efficacy Of Electroconvulsive Therapy: There has been increasing literature regarding the influence of induction agent on the therapeutic efficacy of ECT, which promoted psychiatrists to get involved with the anesthesiologist in making the choice of the induction agent.
Ketamine has intrinsic antidepressant properties as it is an N-methyl-D-aspartate antagonist. It may speed the onset of antidepressant response to ECT but there are mixed evidences of potential development of tolerance due to the repeated use. Administering other induction agent (like thiopental) in alternate treatment sessions with ketamine may prevent the development of tolerance. The enhancement of efficacy early in the course by ketamine, may be particularly advantageous in two situations: (a) in right unilateral ultra-brief ECT where the onset of antidepressant effect is slower, (b) in cases of high suicide risk or high clinical severity (e.g., catatonia), when a very rapid response is required.
Propofol or barbiturates do not have any intrinsic antidepressant properties but combining propofol with Ketamine (ketofol) retains the early antidepressant property of latter, while reducing the adverse effects. This suggests that propofol combined with Ketamine anaesthesia might be the technique of first-choice in patients with depressive disorders undergoing ECT. Adding Dexmedetomidine to this combination (ketofol-dex) has added anti-depressive effect following first ECT session, but not at the end. However, ketofol-dex combination has advantages in the form of increased seizure duration, lower incidence of agitation, more patient satisfaction and acceptable decrease in heart rate and blood pressure when compared to ketofol.
Etomidate may improve major depressive disorder more than sodium thiopental. However, depression is associated with stress-related hypothalamic pituitary adrenal axis dysregulation. This may be of concern regarding the consequences of Etomidate’s unwanted suppression of adrenal function on depression, particularly on the course of illness. However, the findings of Wang et al. in 2011, through measurements of serial cortisol levels at various time points during ECT, found no worrisome reductions with Etomidate.
Complications: ECT is generally considered as safe procedure and complications are rare. Minor complications are usually common and self resolving. However, major complications are rare but may be fatal. Post-ictal agitation is noted in almost 30% patient but use of propofol or Ketofol as an induction agent reduces incidence up to 5%. Pulmonary complications like aspiration, prolonged apnea, pulmonary edema e.g. (0.01%) are very rare but may be life threatening and hence anesthesiologist must be aware of these complications and its management.
Cardiac complications like tachycardia and ventricular ectopics are of minor severity and short lasting also, may not need treatment. On the contrary, major cardiac complications like asystole, complete AV block are rare and may prove fatal. Hence, pre ECT electrocardiogram must be done for all patients especially patients above 50 years.
Table no.1 showing complications of ECT
Minor | Major |
Post ictal agitation (5 %- 30%) | Pulmonary Complications (0.01%) |
Headache, Jaw pain | Cardiac arrhythmia |
Myalgia | Stroke |
Short term memory loss (33%) | Prolonged seizures |
Nausea, vomiting | Death (0.0001%) |
Neuroleptic malignant syndrome (NMS) although rare (0.01% to 0.02%) but is a serious side-effect produced by some antipsychotic drugs like Chlorpromazine and Haloperidol and shares some clinical similarities to malignant hyperthermia (MH). Although evidence is lacking to support NMS and MH having a similar pathophysiology, caution is advised when administering general anaesthesia to patients with NMS. Agents such as sevoflurane, suxamethonium (SCh) or their combination, known to trigger MH should be avoided, though the administration of SCh to patients receiving ECT for NMS is safe. Therefore, in patients with a history of NMS, a more promising method of muscle relaxation is to use Rocuronium-Sugammadex as an alternative to SCh.
Contraindications for ECT are rare but known pheochromocytoma is considered as an absolute contraindications. Relative contraindications include raised ICT, recent MI (3month), severe hypertension, narcotic intolerance, acute glaucoma, changes in cerebral arteries e.g. cerebral aneurysm, angioma are some of relative contraindications for ECT.
Conclusion: ECTs are performed in a set up where an anesthesiologist is in charge of general anaesthesia and a psychiatrist administers ECT. Thus, anesthesiologists and psychiatrists must be aware of not only the physiological responses to ECT and how to modify these but also understand the anesthetic factors that may influence the efficacy of ECT.s
References:
- Surve R, Bansal S, Sriganesh K, Subbakrishna DK, Thirthalli J et al. Incidence and risk factors for oxygen desaturation during recovery from modified electroconvulsive therapy: A prospective observational study. J Anaesthesiol Clin Pharmacol. 2015 Jan-Mar; 31(1): 99-103.
- Sawayama E, Takahashi M, Inoue A, Nakajima K, Kano A, Sawayama T, et al. Moderate hyperventilation prolongs electroencephalogram seizure duration of the first electroconvulsive therapy. J ECT 2008; 24: 195-8.
- Lee M. Anesthesia for electroconvulsive therapy. In: Atlee JL, editor. Complications in Anesthesia. 2nd ed. Philadelphia: Elsevier Health Sciences; 2006. pp. 903–5.
- Pavan KK, Laxmi KK. Anaesthesia for electroconvulsive therapy: An overview with update on its role in potentiating electroconvulsive therapy. Ind J Anesth 2017; 6: 5: 373-80.



