Dialysis Overview and Perioperative Indications for Dialysis

Consultant Nephrologist &
Kidney Transplant Physician
CARE Hospital, Nagpur
nephro@kidneyadvice.net |
www.kidneyadvice.net
Introduction
Dialysis is a process where solute composition of blood is altered by exposing
it to another solution (dialysate) across a semi-permeable membrane. To put it simply,
dialysis is required to achieve metabolic homeostasis when kidneys are almost completely
shut down, either in case of Acute Kidney Injury (AKI) or Chronic Kidney Disease
(CKD).
Stages of CKD
Chronic Kidney Disease progresses through five stages as the GFR declines. These
stages are shown in fig.1.
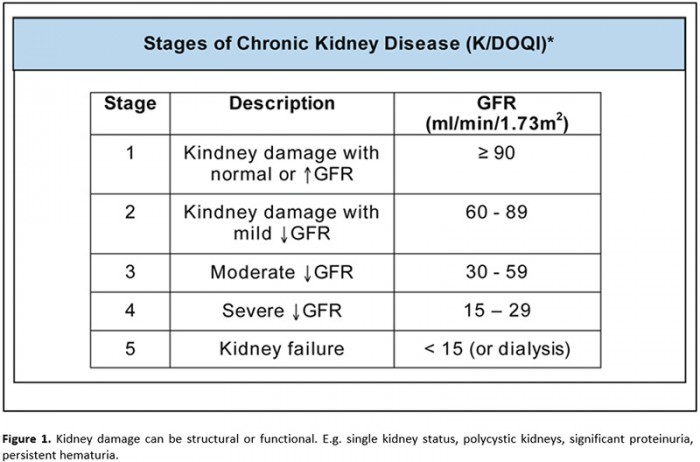
These stages are now further subdivided based on the amount of proteinuria and
some stages are split into two.

Dialysis requirement in CKD
There are no defined time points to initiate dialysis in a CKD patient who is
on a regular follow up. By convention, severe symptoms (e.g. nausea, vomiting, reduced
urine output) in any patient with eGFR less than 15 mL/min/1.73 m2 will
warrant initiation of dialysis. Alternatively, dialysis will also be initiated if
the eGFR is less than 8 – 10 mL/min/1.73 m2 even if most major symptoms
are absent.
Definite indications for initiation of dialysis include symptomatic uremia, pericarditis,
encephalopathy, fluid overload, refractory hyperkalemia, refractory metabolic acidosis
or eGFR < 7 mL/min/1.73 m2.
Dialysis Options
Largely, two variants of dialysis are offered as choice to the patient viz. hemodialysis
and peritoneal dialysis.
Hemodialysis is commonly done in a hospital setting on an OPD or Day-care basis.
Increasingly now, it is done in satellite centres with only emergency back-up from
hospitals. In patients with eGFR less than 2 mL/min/1.73m2, hemodialysis
is recommended to be done at least thrice a week with each session being at least
4 hours of duration. In patients with eGFR more than 2 mL/min/1.73m2,
twice a week hemodialysis can be done with each session being of 6 hours duration.
Figure 3 shows a typical hemodialysis circuit.


Blood flows out from an access ( a large vein from a surgically created AV Fistula
or a hemodialysis catheter), is given a specified flow rate by the ‘blood pump’,
passes through the filter (the dialyzer), where it is ‘treated ‘ with a dialysate
which is a solution that has electrolytes and base at concentrations close to that
of plasma. (Table 1)
The dialyzer (figure 4) is in fact a bundle of capillaries made of semipermeable
membrane dividing itself into a ‘blood compartment’ and ‘dialysate compartment’.
The dialysate flows in a direction opposite to that of the blood.
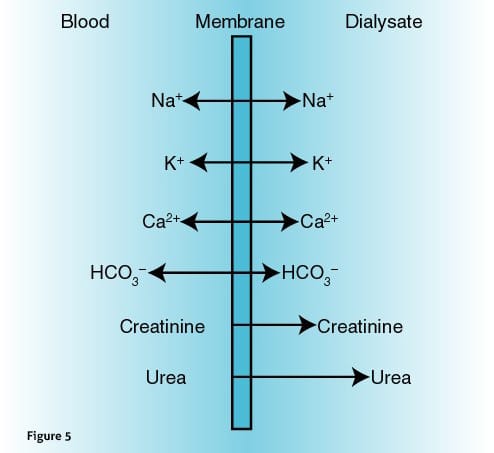
By virtue of the nature of a semipermeable membrane, substances diffuse along
the concentration gradients (figure 5) and therefore, urea, urea derived toxins,
creatinine and potassium will diffuse towards the dialysate and bicarbonate will
diffuse towards the blood compartment. Calcium and Sodium will also follow the gradient
and can be in either direction.
| Solute | Concentration |
|---|---|
| Sodium | 130 – 150 mEq/L |
| Potassium | 0 – 2 mEq/L |
| Calcium | 2.5 – 3.5 mEq/L |
| Magnesium | 0.5 – 0.75 mEq/L |
| Bicarbonate | 30 – 42 mEq/L |
Ultrafiltration (UF) is achieved by application of negative pressure on the dialysate
side resulting in the removal of the precise amount of UF target set. The ‘purified’
blood is returned to the circulation via another ‘downstream’ vein in the AV Fistula
or second port of the hemodialysis catheter. Unfractionated Heparin upto 10,000
units is used in a single hemodialysis session to prevent clotting in the extra-corporeal
circuit.
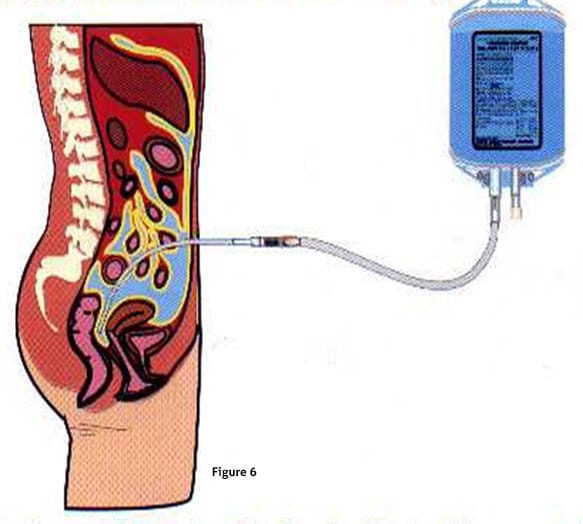
Peritoneal Dialysis
Continuous Ambulatory Peritoneal Dialysis (CAPD) is another alternative that
can be offered to patients requiring dialysis. Here, the access is a surgically
placed catheter inside the peritoneal cavity. A dialysis solution is instilled in
the peritoneal cavity through this catheter. The solution reaches the rich capillary
network lining the parietal peritoneum. An exchange process very similar to that
happening inside the dialyzer of hemodialysis takes place. Here, the capillary wall
acts as the semi permeable membrane.
The Dialysis solution is supplied as a two liter bag. It is a twin bag system
where an empty drain bag is also attached. First step is to empty indwelling fluid
from the previous instillation. After that fresh fluid is instilled through the
same connection. The connections are alternated using clamps. (Fig 7 and Fig 8).


Routinely, 3 to 4 exchanges are performed over 24 hours with a 2 litres dwell
volume and 4 to 6 hours of dwell time. The night exchange is longer (8-10 hrs).
Fluid ‘strength’ is determined by the dextrose concentration in the fluid. Higher
the concentration, more is the ultrafiltration achieved by a particular dwell. This
‘osmotic gradient’ is offset by diffusion of dextrose across the peritoneal membrane.
How much dextrose gets absorbed depends on the ‘exchanging ability’ of the peritoneal
membrane and individuals can be ‘low exchangers’, or ‘high exchangers’. It is easy
to deduce that high exchangers will loose the osmotic gradient faster during the
dwell and therefore end up having less ultrafiltration than low exchangers. Also,
unlike in hemodialysis, the UF achieved is less predictable with CAPD.
Peri-operative assessment of CKD patient.
In a high risk surgery (e.g. cardiac surgery) the morbidity in a patient with
CKD is to the tune of 46%. Perioperative assessment seeks to identify patients at
higher risk of morbidity or mortality. Causes include coronary artery disease, myocardial
dysfunction, electrolyte abnormalities, renal excretion (or the lack of it) of anesthetic
agents, bleeding tendencies and extremes of blood pressure.
The CKD patient falls in class 4 ( or ASA PS4) of the American Society of Anesthesiologist’s
classification of preoperative ‘Physical State’ (PS) by virtue of the disease being
a “constant threat to life”. A patient for an elective procedure with an ASA grade
of 3 or 4 needs a consultation with a senior anaesthetist as far before the proposed
surgery as is possible. This will enable the patient’s physical condition to be
optimised with better post-operative outcome and will reduce the chance of ‘on the
day’ cancellation due to being medically unfit.
Depending on the eGFR, the extent of dissection in a given surgery and the operative
time, presence of CKD can affect in following ways:
- Higher intraoperative and/or post-operative bleeding.
- Increase in Hospital Stay and/or ICU stay.
- Longer time for wound healing.
- Electrolyte disturbances.
- Worsening of Renal Functions. Sometimes dialysis requirement.
- Increased mortality.
Dialysis in the peri-operative period.
Dialysis is often required in the perioperative period to optimize the surgical
risk, reduce bleeding tendencies, correct metabolic disturbances or to achieve euvolemia
in an oliguric or anuric patient. The decision is easier in CKD stage V patients
who are already on maintenance dialysis. A routine session of hemodialysis (heparin
free) should be done preferably upto 8 hours prior to surgery. This duration allows
fluids, electrolytes and acid-bases to “equilibrate” prior to induction of anesthesia.
In an emergency situation, attention should focus on acid-base abnormalities, potassium
levels and fluid overload. With hyperkalemia, one can go ahead with the surgery
if ECG shows no changes of hyperkalemia. If such changes are seen, a short HD of
2 hours duration is sufficient to correct hyperkalemia.
In patients who are on CAPD, exchanges should continue as before. An additional
exchange per day starting one week prior to the surgery is recommended. Exchanges
should be stopped just before the surgery and patient should be shifted with a ‘dry’
abdomen for the surgery. For abdominal surgery in a CAPD patient, temporary switch
to hemodialysis is advisable.
It goes without stating that euvolemia is desirable prior to induction of anesthesia.
In hemodialysis patients, this can be achieved effectively in a 2 – 4 hours session.
In patients on CAPD, faster low volume exchanges can markedly increase UF. A common
practice in a fluid overloaded CAPD patient is to perform one hourly exchanges of
1 litre 2.5% fluid till a desired volume of UF is achieved.
In patients with CKD stage IV (i.e eGFR less than 30 mL/min/1.73 m2) undergoing
a major surgery, dialysis can be avoided in the pre-operative period if Arterial
Blood gases (ABG) are normal, electrolytes are normal, there are no bleeding tendencies
and BUN is less than 75 mg/dL. One must keep in mind that the bleeding tendencies
in uremia is a result of platelet dysfunction which can only be well reflected in
clotting time and bleeding time. However, there are studies contradicting any correlation
of bleeding time with occurrences of excessive surgical bleeding and therefore,
checking bleeding time is currently not a recommendation. In patients with elevated
bleeding time and active excessive bleeding, aggressive dialysis may correct the
bleeding tendencies. Other options include Desmopressin (IV or intranasal), Cryoprecipitate
and elevation of hematocrit by blood transfusions.
In patients with eGFR less than 20 mL/min/1.73m2, decision to dialyze prior to
surgery should be based on volume status, electrolytes and acid base status. In
patients with eGFR close to 15 mL/min/1.73 m2 who are not on dialysis, it is safer
to do dialysis prior to surgery irrespective of the above factors.
Post operatively, in patients who have been on maintenance hemodialysis, it should
be continued on alternate day basis. However it can be more frequent depending on
volume status and electrolytes. Similarly CAPD (except in abdominal surgeries) should
also be continued as before.
Summary and Conclusions.
CKD is a unique population affecting the surgical outcomes. Their requirements
are also unique including the potential requirement for dialysis. Dialysis, whether
HD or CAPD, helps achieve euvolemia, metabolic homeostasis and improves outcomes.
Increasing number of ESRD patients now undergo major surgeries including CABG, successfully.
Dialysis should be done pre-operatively to improve surgical fitness and postoperatively
to reduce mortality and morbidity.



